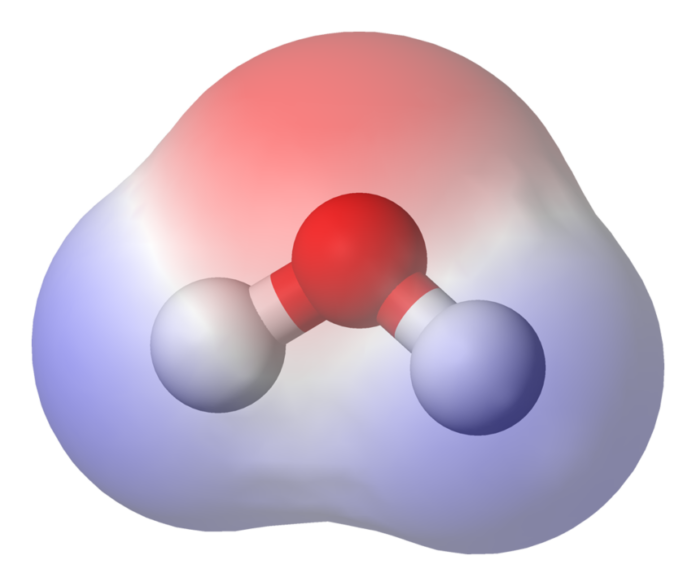
Molecular polarity is a key concept in chemistry that explains many physical and chemical properties of substances. It influences solubility, boiling and melting points, reactivity, and intermolecular interactions. This concept revolves around the distribution of electrical charge within a molecule and determines whether the molecule is polar or nonpolar.
What Is Molecular Polarity?
Molecular polarity refers to the uneven distribution of electron density in a molecule, resulting in a separation of electric charge. This separation creates a dipole moment, which is a vector quantity with both magnitude and direction.
A molecule’s polarity depends on two factors:
- Bond polarity: The polarity of individual chemical bonds within the molecule.
- Molecular geometry: The three-dimensional arrangement of atoms in the molecule.
A polar molecule has an asymmetrical distribution of charges, while a nonpolar molecule has a symmetrical distribution, leading to no net dipole moment.
Bond Polarity
Bond polarity arises from the difference in electronegativity between two bonded atoms. Electronegativity is the ability of an atom to attract shared electrons in a bond. When two atoms have different electronegativities, the electrons are more attracted to the more electronegative atom, creating a partial negative charge (\(\delta^-\)) on that atom and a partial positive charge (\(\delta^+\)) on the less electronegative atom.
The degree of bond polarity is determined by the difference in electronegativity:
- Nonpolar covalent bond: Electronegativity difference is less than 0.5. Electrons are shared equally.
- Polar covalent bond: Electronegativity difference is between 0.5 and 1.7. Electrons are unequally shared.
- Ionic bond: Electronegativity difference is greater than 1.7, leading to complete electron transfer.
For example:
- In a water molecule (\(H_2O\)), the oxygen-hydrogen bond is polar due to oxygen’s higher electronegativity compared to hydrogen.
- In a methane molecule (\(CH_4\)), the carbon-hydrogen bond is considered nonpolar because the electronegativity difference is minimal.
Molecular Geometry and Polarity
While bond polarity contributes to molecular polarity, the overall shape of the molecule determines whether individual bond polarities cancel each other out or create a net dipole moment. Molecular geometry is predicted using the Valence Shell Electron Pair Repulsion (VSEPR) theory.
Symmetrical Molecules
Molecules with symmetrical shapes tend to be nonpolar because their bond polarities cancel out. Examples include:
- Carbon dioxide (\(CO_2\)): Linear geometry with two polar bonds pointing in opposite directions, resulting in no net dipole moment.
- Boron trifluoride (\(BF_3\)): Trigonal planar geometry with equal bond polarities evenly distributed, making the molecule nonpolar.
Asymmetrical Molecules
Molecules with asymmetrical shapes are typically polar because the bond polarities do not cancel out. Examples include:
- Water (\(H_2O\)): Bent geometry with two polar bonds pointing in different directions, creating a net dipole moment.
- Ammonia (\(NH_3\)): Trigonal pyramidal geometry with polar bonds, resulting in a net dipole moment.
Dipole Moment
The dipole moment (\(\mu\)) is a quantitative measure of molecular polarity. It is expressed as:
\(\mu = q \cdot d\)Where:
- \(q\) = Magnitude of charge
- \(d\) = Distance between charges
The dipole moment is measured in Debye units (D). Molecules with a higher dipole moment are more polar.
For instance:
- Water (\(H_2O\)) has a dipole moment of approximately 1.85 D, making it highly polar.
- Carbon dioxide (\(CO_2\)) has a dipole moment of 0 D because it is nonpolar.
Polar and Nonpolar Molecules
Polar Molecules
- Have an uneven charge distribution.
- Exhibit strong intermolecular forces, such as hydrogen bonding and dipole-dipole interactions.
- Are generally soluble in polar solvents (e.g., water).
Examples:
- Water (\(H_2O\)): Highly polar due to its bent shape and electronegative oxygen atom.
- Hydrogen chloride (\(HCl\)): Polar because of the significant electronegativity difference between hydrogen and chlorine.
Nonpolar Molecules
- Have a uniform charge distribution.
- Exhibit weaker intermolecular forces, such as London dispersion forces.
- Are generally soluble in nonpolar solvents (e.g., hexane).
Examples:
- Methane (\(CH_4\)): Nonpolar due to its symmetrical tetrahedral shape.
- Oxygen (\(O_2\)): Nonpolar because it consists of two identical atoms sharing electrons equally.
Applications of Molecular Polarity
Molecular polarity plays a crucial role in various scientific and industrial processes:
Solubility and Solutions
- Polar molecules dissolve in polar solvents, while nonpolar molecules dissolve in nonpolar solvents. This principle is summarized by the phrase “like dissolves like.”
- For example, salt (\(NaCl\)) dissolves in water but not in oil due to water’s polarity.
Boiling and Melting Points
- Polar molecules have higher boiling and melting points than nonpolar molecules because of stronger intermolecular forces.
- For instance, water (\(H_2O\)) has a higher boiling point than methane (\(CH_4\)).
Biological Systems
- Polarity is essential in biological systems, influencing protein folding, membrane formation, and enzyme activity.
- The polar nature of water makes it an excellent solvent for transporting nutrients and waste in living organisms.
Industrial Applications
- Molecular polarity is critical in the production of emulsifiers, which stabilize mixtures of polar and nonpolar substances, such as oil and water.
- It is also significant in chromatography techniques, where separation of compounds is based on polarity differences.
Factors Affecting Polarity
Several factors can influence molecular polarity:
- Electronegativity Differences: Greater differences result in stronger bond polarity.
- Molecular Shape: Symmetrical shapes reduce polarity, while asymmetrical shapes enhance it.
- Presence of Lone Pairs: Lone pairs on central atoms often create asymmetry, increasing polarity.
Understanding molecular polarity is essential for explaining the behavior and properties of substances. From the solubility of compounds to biological interactions and industrial applications, polarity is a cornerstone of chemistry. By analyzing bond polarity and molecular geometry, scientists can predict and manipulate the physical and chemical properties of molecules to suit various applications. Mastery of this concept not only enhances knowledge of fundamental chemistry but also provides tools for solving real-world problems.